Urgent message: Clinical evaluation that includes pretest probability tools and judicious use of diagnostic tests is a requirement for patients who present in the urgent care setting with symptoms suggestive of VTE.
MELVIN LEE, MD, CCFP, RMC
Early diagnosis of venous thromboembolism (VTE) is important to prevent the morbidity and mortality associated with it.Introduction
VTE is subdivided into pulmonary embolism and deep venous thrombosis (DVT). DVT is most common in lower extremities. Those involving deep veins proximal to the knee are associated with an increased risk of pulmonary embolism (PE), whereas if only the calf veins are involved, PE is less likely but risk of development of postthrombotic syndrome is higher. Postthrombotic (postphlebitic) syndrome refers to the development of chronic venous symptoms and/or signs secondary to DVT and can include pain, venous dilation, edema, pigmentation, skin changes, and venous ulcers. Upper extremity DVT is uncommon and will not be discussed in this article.
The annual incidence of VTE in the United States is 600,000 cases and is increasing with the aging of the population.1 Even with prompt diagnosis and treatment, 10% of VTEs are rapidly fatal and another 5% of patients die soon thereafter. Twenty-six percent of undiagnosed and untreated patients with PE will have a subsequent fatal embolic event, whereas another 26% will have a nonfatal recurrent embolic event that can eventually be fatal.2-4 Therefore early diagnosis to prevent mortality and morbidity associated with VTE is paramount.
DVT is the presence of coagulated blood in one of the deep veins that return blood to the heart. Symptoms such as pain and swelling are often nonspecific or absent. If left untreated, a clot may become fragmented or dislodge and migrate to obstruct the pulmonary artery, causing potentially life-threatening PE.
Lower-extremity DVT is the most common venous thrombosis, with a prevalence of over 1 case per 1,000 population. In addition, proximal DVT is the underlying source of 90% of acute PEs, which cause 25,000 deaths per year in the United States.3-6
The United States-wide prevalence of venous throm- boembolism (VTE) increased by 33.1% from 2002 to 2006. During the same period, the annual incidence increased with age as well.7 These data confirm that VTE remains a major healthcare burden in the United States, particularly among the elderly, and highlight a contin- uing increase in incidence of the disease.
Risk Factors for Venous Thromboembolism
Risk factors for venous thromboembolic disease include increasing age, immobilization, surgery, trauma, malignancy, pregnancy, estrogenic medications (such as oral contraceptive pills, hormone therapy, tamoxifen), congestive heart failure, hyperhomocystinemia, diseases that alter blood viscosity (such as polycythemia, sickle cell disease, and multiple myeloma), and inherited thrombophilia. About 75% of patients with venous thromboembolic disease have at least one established risk factor, and half of all cases of DVT occur in hospitalized patients or nursing home residents.8 Inherited thrombophilias can be identified in 24% to 37% of patients with DVT and in the majority of patients with familial thrombosis. 9,10 Risk factors for VTE can be divided into two groups—hereditary and acquired—as shown in Table 1.11,12
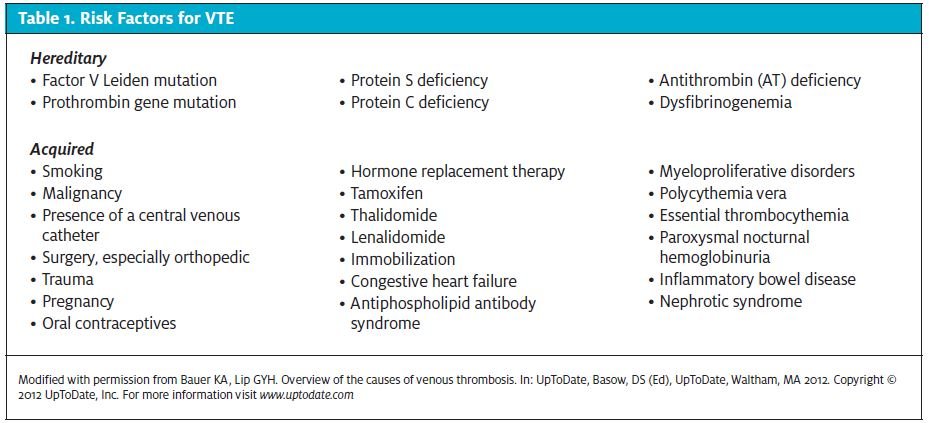
Approximately 20% to 25% of all new cases are asso- ciated with the presence of an active malignancy, 20% are associated with trauma, 20% are associated with a concurrent or recent medical hospitalization, and 25% are idiopathic, with no obvious provoking risk factor.6 DVT of the lower limb normally starts in the calf veins. About 10% to 20% of thromboses extend proximally, and another 1% to 5% of patients go on to develop fatal PE.
Pathophysiology
The formation of a pulmonary embolus from a clot migrating from the venous circulation was first described in 1856 by Dr. Rudolph Virchow and was later called Virchow’s Triad.13 The triad has 3 factors:
- Stasis
- Vessel damage
- Hypercoagubility14
The triad is used to determine etiology and risk of venous thrombosis, especially DVT. At least 2 of the 3 factors have to be present to increase a patient’s risk of DVT. Blood clots tend to form in connection with venous valves in the veins. A clot can propagate and grow proximally across the lumen. Most clots start in the calf veins and propagate proximally. Blood clots associated with pregnancy or hip arthroplasty can start in the pelvic veins. Most blood clots that develop in the deep venous system of the calf begin to form just above and behind a venous valve.15,16
As soon as a clot begins to form, the fibrinolytic system begins to dissolve fibrin blood clots. The D-dimer antigen is a unique marker of fibrin degradation that is formed by the sequential action of 3 enzymes: thrombin, factor XIIIa, and plasmin. Once the plasmin degrades the crosslinked fibrin, the D-dimer antigen is exposed and can be measured in the laboratory. A normal level of D-dimer provides strong evidence against acute thrombosis.
Clinical Presentation and Diagnosis
Deep Vein Thrombosis
Patients with DVT may present with pain, erythema, tenderness, and edema of one limb. In lower-limb DVT, the calf is usually swollen. Other causes of leg swelling, erythema, and tenderness include a ruptured Baker’s cyst and infective cellulitis. Ruptured Baker’s cysts are commonly associated with a history of osteoarthritis and rheumatoid arthritis. Patients who have cellulitis usually have signs of redness, warmth and swelling of the skin. A portal of entry may be determined.
On objective exam, any unilateral leg and thigh swelling can be assessed by measuring the circumference of the leg 10 cm below the tibial tuberosity and 10 cm to 15 cm above the upper edge of the patella. There may be pitting edema present, worse on the affected side, and new dilated collateral superficial veins. There may be localized tenderness along the path of the deep veins. When massive, the swelling can obstruct not only venous outflow but arterial inflow of the affected leg, leading to phlegmasia cerulea dolens (literally means swollen, blue and painful) due to ischemia.
In many cases, the diagnosis of cellulitis or a muscu- loskeletal injury is straightforward, but the provider may have to rule out a DVT if the diagnosis is uncertain.
Pulmonary Embolism
The history and physical are not usually sufficient to confirm or rule out PE. Chest pain and dyspnea are the most common presenting complaints. Patients may also present with cough and hemoptysis. DVT may not be suspected clinically, but its presence, along with thrombotic risk factors, will make the diagnosis of PE more likely. A similar clinical probability model to that for DVT has been developed for PE.
Physical examination is often unremarkable. The most common signs are tachypnea, rales, and tachycardia.
Respiratory examination is usually normal. Crackles or decreased breath sounds may be heard. Decreased osygen saturation may occur. The cardiac examination may reveal right heart failure is present with elevated jugular venous pressure, sternal heave, and an accentuated pulmonic component to the second heart sound. Hypotension and hypoxemia may indicate a massive PE.Unilateral swelling of the calf or thigh may be evident if DVT is present.17,18
Initial tests such as chest x-ray, ECG, and ABG cannot definitively establish or eliminate PE as a diagnosis.
ECG findings suggestive of PE include tachycardia (37%), new right axis deviation (5%), new right bundle branch block (11%), and S wave in lead I, Q wave with T wave inversion in lead III (12%) (findings only suggestive of PE not diagnostic). Those associated with poor prognosis include atrial arrhythmias, right Bundle Branch Block, inferior Q waves, and precordial T wave inversion and ST segment changes.19,20
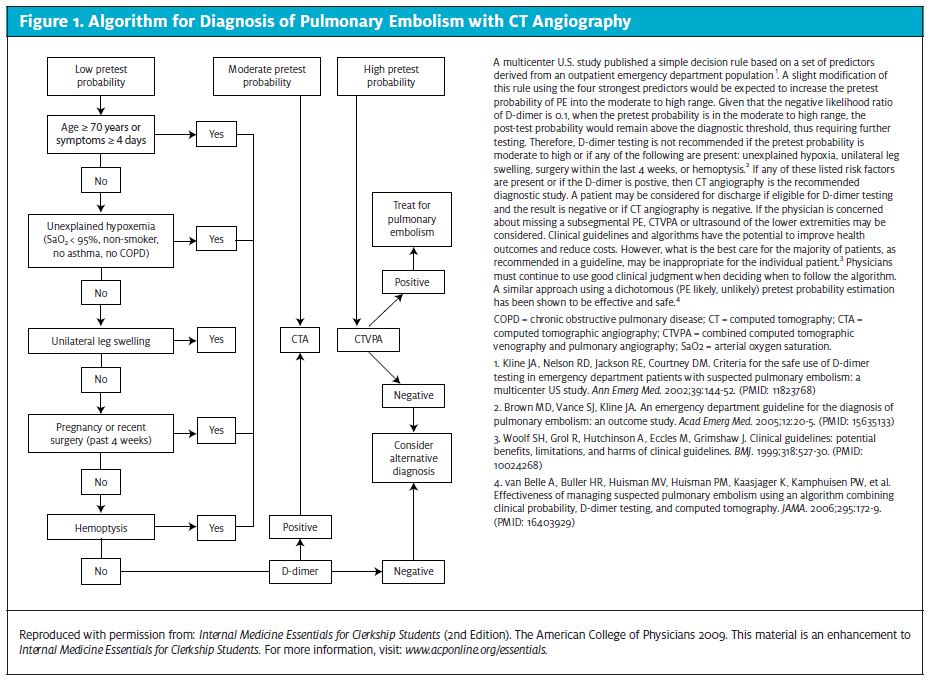
Chest x-ray abnormalities are only suggestive and not diagnostic. They include Fleischner sign: prominent central pulmonary artery (20%); Westermark sign: oligemia in the PE’s area of distribution (11%); Hampton Hump: pleural based areas of increased opacity in the distribution of the PE (27%); and band atelectasis and elevation of the hemi diaphragm.21 Clinical probability using the modified Wells scoring system should be calculated. If the patient is at a low risk of PE, a D-dimer should be measured by rapid quantitative ELISA. If this test is not ele-vated, PE is effectively ruled out without further testing. An algorithm illustrating use of D-dimer testing and ultrasound in patients with suspected DVT is available at http://www.ncbi.nlm.nih.gov/pmc/arti- cles/PMC1609160/figure/f2-21/.
Figure 1 illustrates diagnosis of PE with CT angiography.
Prediction Score
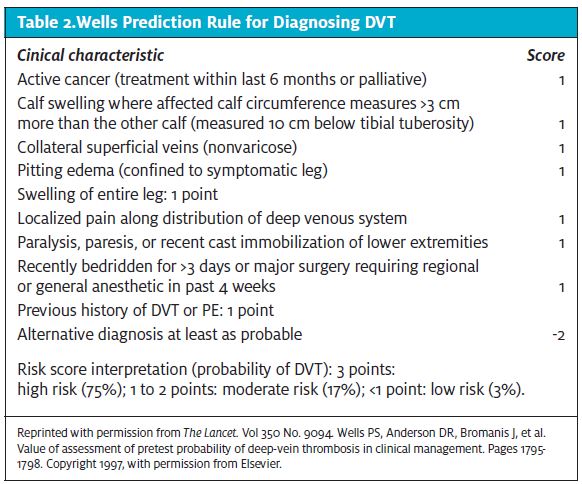
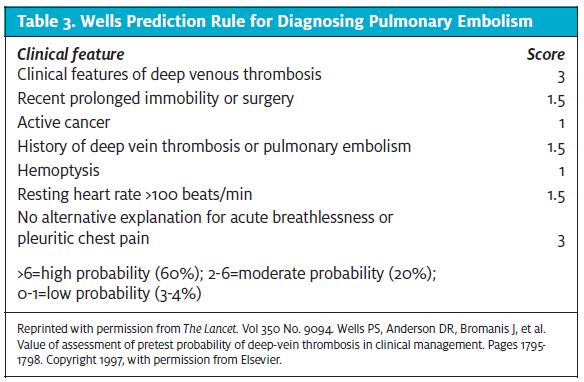
Current evidence supports using a clin- ical prediction rule to establish the pretest probability of VTE. Providers should use the Wells prediction rule to determine the probability of DVT (Table 2) and PE (Table 3) before per- forming and interpreting other diagnos- tic tests. One caveat is that the Wells prediction rule is more accurate in younger patients without comorbidities or a history of VTE. 22-24 Patients with a low pretest probability and a negative D-dimer have a very low likelihood of VTE and that reduces the need for further imaging.25
D-Dimer Testing
In patients with a low pretest probability of DVT or PE as defined by the Wells prediction criteria, the provider can obtain a high-sensitivity D-dimer as a reasonable option, and if negative, this indicates a low likelihood of VTE. A negative highly sensitive D-dimer test largely excludes the diagnosis of DVT and PE in younger patients whose symptoms are of short duration and whose pretest probability score is low. The negative predictive value of the D-dimer is lower in older patients, those with recent surgery, trauma, cancer and those who are postpartum.
It is important to note that the sensitivity of the D- dimer is dependent on the assay method (quantitative enzyme-linked immunoassay methods are more sensi- tive than semiquantitative latex agglutination methods) and the assay cutoff level. At present, ELISA and advanced turbidimetric D-dimer tests are highly sensi- tive assays (sensitivity 96% and 100%) that are practi- cal to use. The sensitivity of D-dimer assays varies, so providers need to be informed of the type of D-dimer assay being used.26-29
The rapid D Dimer tests that are used in urgent care are qualitative or quantitative tests and are CLIA waived. Some studies are reporting that a qualitative D Dimer assay at the point of care has a similar diagnostic accuracy to the lab based quantitative D-dimer test.
An elderly patient receiving a positive D-dimer test result faces stress and delays that could have been avoided if the physician instead had ordered more definitive radiologic imaging as the frontline test. For cases in which the radiologic evaluation confirms VTE, the patient faced delays before receiving treatment for this potentially life-threat- ening condition. In recognition of this problem, joint guide- lines from the American Academy of Family Physicians and American College of Physicians note that D-dimer testing alone may be insufficient to rule out VTE in older patients.26,27 In addition, the authors of several research studies have urged clinicians to consider noninvasive radiologic examinations (such as venous ultrasonography) as a first-line test for diagnosing VTE in older patients.27-30
Ultrasonography
Compression ultrasonography is the non-invasive investigation of choice for the diagnosis of clinically suspected DVT and a moderate or high clinical pretest probability according to the Wells criteria. It is highly sensitive and specific in detecting proximal DVT although less accurate for isolated calf DVT, so negative ultrasonography cannot rule out DVT in these patients.31-35
In patients with suspect- ed calf vein thrombosis and a negative compression ultrasound result, ultrasonography or venography should be repeated, as well as in patients with suspect- ed proximal DVT but whose ultrasonography results are inadequate. Contrast venography is still considered the definitive test to rule out DVT.
Venography
Ultrasound is not as accurate at detecting clots as the diag- nostic standard test, ascending venography, which should be used if the probability of DVT is high and ultra- sound is normal.
If venography cannot be performed, a repeat ultra- sound of the proximal deep venous system is recommend- ed 1 week after the initial ultrasound.36
Helical Computed Tomography
Recent evidence suggests that helical computed tomog- raphy (CT) may have a higher specificity and sensitivi- ty compared with conventional pulmonary arteriogra- phy for the diagnosis of pulmonary embolism. Howev- er, in patients who have a high pretest probability of PE and a negative CT scan, further imaging studies (such as ventilation-perfusion scan, multidetector helical CAT) are needed. For those patients, evidence suggests that CT alone may not be sensitive enough to exclude PE. A single or sequential ultrasonography assessment of the lower extremities or pulmonary angiography may be warranted.37-40
VTE Treatment
Proximal DVT is considered to be of more importance clinically, because it is more commonly associated with PE. One study showed that in up to 56% of cases of proximal DVT, silent PE has already occurred by the time that the patient was seen.41
Distal calf vein thrombosis is felt to be of lesser clinical importance than proximal vein thrombosis and its optimal diagnosis and treatment have yet to be defined. If anticoagulation is not administered for isolated asymptomatic distal venous thrombosis, serial noninvasive studies of the lower extremity may be performed over the next 10 to 14 days to assess for proximal extension of the thrombus, which has a higher incidence of PE.
- the deep veins in the legs;
- Minimize the risk of acute recurrent DVT or PE; and
- Minimize the likelihood of developing postthrom- botic
Criteria for referral for consideration of ED referral include:
- Suspected PE
- Positive ultrasound
- Pretest probability intermediate or higher
Inpatient Treatment
Low-molecular-weight heparin (LMWH) is superior to unfractionated heparin for initial treatment of DVT because it reduces mortality rates and the risk of major bleeding during initial therapy.42-45 Therefore, the Amer- ican College of Physicians (ACP) and the American Academy of Family Physicians (AAFP) recommend that LMWH be used for initial inpatient treatment of DVT. Unfractionated heparin or LMWH is appropriate for initial treatment of patients with PE.46
Outpatient Treatment
In stable patients with symptoms only of a DVT for whom the required support services are in place, outpa- tient treatment of VTE with LMWH is as safe as inpa- tient treatment and is cost-effective. It is recommended that heparin be started at the same time as warfarin. The initial dose of warfarin should not exceed 5 mg . The heparin should be given for a minimum of 5 days and continued until the INR is 2.0 for two consecutive days. The therapeutic range for INR is 2.0 to 3.0.
Anticoagulation Therapy
The ACP and AAFP recommend that anticoagulation therapy be maintained for 3 to 6 months in patients with VTE secondary to reversible risk factors. For patients with recurrent VTE, anticoagulation therapy should be continued for more than 12 months. The exact duration of anticoagulation therapy is not fully understood in patients with idiopathic or recurrent VTE, but extended-duration therapy can provide sub- stantial benefit to these patients. Physicians should weigh the harms, benefits, and patient preferences when deciding the duration of anticoagulation therapy.
Long-Term Management of VTE
Most patients are treated with Coumadin long-term but LMWH is comparable to oral anticoagulation ther- apy in select patients with VTE, and it may be useful in treating patients whose International Normalized Ratio is difficult to control. Therefore, the ACP and the AAFP recommend the use of LMWH as a safe and effective therapy for the long-term treatment of VTE. In addition, LMWH may be more effective than oral anticoagulation therapy in patients with cancer.
References
- Anderson FA Jr, Wheeler HB, Goldberg RJ, et A population-based perspective of the hospital incidence and case-fatality rates of deep vein thrombosis and pulmonary embolism. The Worcester DVT Study. Arch Intern Med. 1991;151(5):933-938.
- Barritt DW, Jordan Anticoagulant drugs in the treatment of pulmonary embolism. A controlled trial. Lancet. 1960;1(7138):1309-1312.
- Heit JA, Melton LI 3rdLohse CM, et Incidence of venous thromboembolism in hospi- talized patients vs community residents. Mayo Clin Proc. 2001;76(11):1102-1110.
- Kearon Natural history of venous thromboembolism. Circulation. 2003;107(23)(suppl 1):122-130.
- Heit JA, Silverstein MD, Mohr DN, et The epidemiology of venous thromboembolism in the community. Thromb Haemost. 2001;86:452-463
- White The epidemiology of venous thromboembolism. Circulation. 2003;107(23 suppl 1):14-18
- Deitelzweig SB, Johnson BH, Lin J, Schulman Prevalence of clinical venous thromboem- bolism in the USA: current trends and future projections. Am J Hematol. 2011;86(2):217-220.
- Heit JA, O’Fallon WM, Petterson TM, Lohse CM, Silverstein MD, Mohr DN, et Rel- ative impact of risk factors for deep vein thrombosis and pulmonary embolism: a popu- lation-based study. Arch Intern Med. 2002;162:1245–1248.
- Mateo J, Oliver A, Borrell M, Sala N, Fontcuberta Laboratory evaluation and clini- cal characteristics of 2,132 consecutive unselected patients with venous thromboem- bolism—results of the Spanish Multicentric Study on Thrombophilia (EMET-Study). Thromb Haemost. 1997;77:444–451.
- Bauer The thrombophilias: well-defined risk factors with uncertain therapeutic implications. Ann Intern Med. 2001;135:367–373.
- Bauer KA, Lip Overview of the causes of venous thromboembolism. UpToDate. http://www.uptodate.com/contents/overview-of-the-causes-of-venous- thrombosis?source=search_result&search=venous+thromboembolism&selectedTitle=1~150 Accessed November 20, 2012.
- Pomp ER, Rosendaal FR, Doggen Smoking increases the risk of venous thrombosis and acts synergistically with oral contraceptive use. Am J Hematol. 2008;83(2):97-102.
- Bagot CN, Arya Virchow and his triad: A question of attribution. Br J Haematol. 2008;143(2):180-190.
- Malone PC, Agutter The aetiology of deep venous thrombosis. Q J Med. 2006;99:581- 593.
- Turpie AG, Chin BS, Lip Venous thromboembolism: pathophysiology, clinical fea- tures, and prevention. BMJ. 2002;325:887-890.
- Cogo A, Lensing AW, Prandoni P, et Distribution of thrombosis in patients with symp- tomatic deep vein thrombosis: implications for simplifying the diagnostic process with com- pression ultrasound. Arch Intern Med. 1993;153:2777-2780.
- Stein PD, Terrin ML, Hales CA, et Clinical laboratory, roentgenographic, and electro- cardiographic findings in patients with acute pulmonary embolism and no pre-existing car- diac or pulmonary disease. Chest. 1991;100(3):598-603.
- Stein PD, Saltzman HA, Weg Clinical characteristics of patients with acute pulmonary embolism. Am J Cardiol. 1991;68(17):1723-1724.
- Geibel A, Zehender M, Kasper W, Olschewski M, Klima C, Konstantinides Prognos- tic value of the ECG on admission in patients with acute major pulmonary embolism. Eur Respir J. 2005;25(5):843.
- Ferrari E, Imbert A, Chevalier T, Mihoubi A, Morand P, Baudouy The ECG in pulmonary embolism. Predictive value of negative T waves in precordial leads—80 case reports. Chest. 1997;111(3):537.
- Worsley DF, Alavi A, Aronchick JM, et Chest radiographic findings in patients with acute pulmonary embolism: observations from the PIOPED Study. Radiology. 1993;189:133- 136.
- Dryjski M, O’Brien-Irr MS, Harris LM, Hassett J, Janicke Evaluation of a screening pro- tocol to exclude the diagnosis of deep venous thrombosis among emergency department patients. J Vasc Surg. 2001;34:1010–1015.
- Anderson DR, Wells PS, Stiell I, et Management of patients with suspected deep vein thrombosis in the emergency department: combining use of a clinical diagnosis model with D-dimer testing. J Emerg Med. 2000;19:225–230.
- Wells PS, Anderson DR, Bormanis J, et Value of assessment of pretest probability of deep-vein thrombosis in clinical management. Lancet. 1997;350:1795–1798.
- Fancher TL, White RH, Kravitz Combined use of rapid D-dimer testing and estima- tion of clinical probability in the diagnosis of deep vein thrombosis: systematic review. BMJ. 2004;329:821.
- Qaseem A, Snow V, Barry P, et al; Joint American Academy of Family Physicians/Amer- ican College of Physicians Panel on Deep Venous Thrombosis/Pulmonary Cur- rent diagnosis of venous thromboembolism in primary care: a clinical practice guideline from the American Academy of Family Physicians and the American College of Physicians. Ann Fam Med. 2007;5(1):57-62.
- Stein PD, Hull RD, Patel KC, et D-dimer for the exclusion of acute venous thrombo- sis and pulmonary embolism: a systematic review. Ann Intern Med. 2004;140(8):589-602.
- Righini M, Le Gal G, Perrier A, et The challenge of diagnosing pulmonary embolism in elderly patients: influence of age on commonly used diagnostic tests and strategies. J Am Geriatr Soc. 2005;53(6):1039-1045.
- Antonelli F, Villani L, Masotti L, et Ruling out the diagnosis of venous thromboem- bolism in the elderly: is it time to revise the role of D-dimer? Am J Emerg Med. 2007;25(6):727-728.
- Schutgens RE, Haas FJ, Biesma Reduced efficacy of clinical probability score and D-dimer assay in elderly subjects suspected of having deep vein thrombosis. Br J Haema- tol. 2005;129(5):653-657.
- White RH, McGahan JP, Daschbach MM, Hartling Diagnosis of deep-vein thrombo- sis using duplex ultrasound. Ann Intern Med. 1989;111:297–304.
- Becker DM, Philbrick JT, Abbitt Real-time ultrasonography for the diagnosis of lower extremity deep venous thrombosis. The wave of the future? Arch Intern Med. 1989;149:1731–1734.
- Cogo A, Lensing AW, Wells P, Prandoni P, Buller Noninvasive objective tests for the diagnosis of clinically suspected deep-vein thrombosis. Haemostasis. 1995;25:27–39.
- Kearon C, Julian JA, Newman TE, Ginsberg Noninvasive diagnosis of deep venous thrombosis. McMaster Diagnostic Imaging Practice Guidelines Initiative. Ann Intern Med. 1998;128:663–677.
- Gottlieb RH, Widjaja J, Tian L, Rubens DJ, Voci Calf sonography for detecting deep venous thrombosis in symptomatic patients: experience and review of the literature. J Clin Ultrasound. 1999;27:415–420.
- Gibson NS, Schellong SM, El Kheir DY, et Safety and sensitivity of two ultrasound strategies in patients with clinically suspected deep venous thrombosis: a prospective man- agement study. J Thrombo Haem. 2009;7(12):2035-2041.
- Rathbun SW, Raskob GE, Whitsett Sensitivity and specificity of helical computed tomography in the diagnosis of pulmonary embolism: a systematic review. Ann Intern Med. 2000;132:227–232.
- Winer-Muram HT, Rydberg J, Johnson MS, et Suspected acute pulmonary embolism: evaluation with multi-detector row CT versus digital subtraction pulmonary arteriography. Radiology. 2004;233:806–815.
- Roy PM, Colombet I, Durieux P, et Systematic review and meta-analysis of strate- gies for the diagnosis of suspected pulmonary embolism. BMJ. 2005;331:259.
- Quiroz R, Kucher N, Zou KH, et Clinical validity of a negative computed tomogra- phy scan in patients with suspected pulmonary embolism: a systematic review. JAMA. 2005;293:2012–2017.
- Girard P, Decousus M, LaPorte S, et Diagnosis of pulmonary embolism in patients with proximal deep vein thrombosis: specificity of symptoms and perfusion defects at base- line and during anticoagulant therapy. Am J Respir Crit Care Med. 2001;164:1033-1037.
- Gould MK, Dembitzer AD, Sanders GD, Garber Low-molecular-weight heparins compared with unfractionated heparin for treatment of acute deep venous thrombosis. A cost-effectiveness analysis. Ann Intern Med. 1999;130:789-799.
- Green D, Hirsh J, Heit J, Prins M, Davidson B, Lensing Low molecular weight heparin: a critical analysis of clinical trials. Pharmacol Rev. 1994;46:89-109.
- Hettiarachchi RJ, Prins MH, Lensing AW, Buller Low molecular weight heparin ver- sus unfractionated heparin in the initial treatment of venous thromboembolism. Curr Opin Pulm Med. 1998;4:220-225.
- Hirsh J, Siragusa S, Cosmi B, Ginsberg Low molecular weight heparins (LMWH) in the treatment of patients with acute venous thromboembolism. Thromb Haemost. 1995;74:360-363.
- Snow V, Qaseem A, et Management of Venous Thromboembolism: A Clinical Prac- tice Guideline from the American College of Physicians and the American academy of Fam- ily Physicians. Annal Fam Med. Vol. 5, No. 1. Jan/Feb 2007
